VAERS reports of adverse reactions after Covid vaccination total more than 1.56 million and include 36,324 deaths
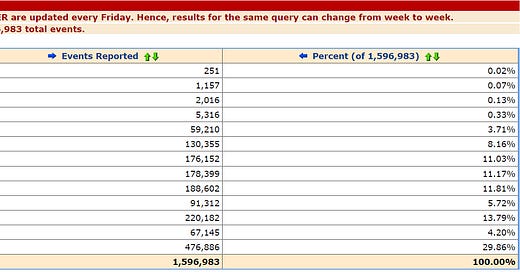
The database of the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) in the US listed 1,596,983 adverse event reports after Covid vaccination as of September 29, 2023, but 35,931 reports were described as ‘no adverse event’.
According to the data as of September 29, 36,324 deaths after Covid vaccination have been reported to VAERS.
The statistics include adverse reactions reported after administration of the bivalent boosters.
Including adverse reactions reported after administration of the bivalent boosters 7,302,481 individual-symptom events are listed (many reports include more than one symptom).
There are 79,000 reports described as vaccination failure and 73,850 listed as “drug ineffective”. There are 39,242 reports of an expired product being administered, 36,348 reports of a product storage error, 30,497 reports of an inappropriate schedule of product administration, 26,563 reports of off-label use, 13,688 reports of a product being administered to patient of an inappropriate age, 11,484 reports of an incorrect dose being administered, 9,530 reports of an extra dose being administered, 8,433 reports of an incorrect product formulation being administered, and 7,610 reports of a “poor quality product” being administered.
VAERS now has a separate category, ‘COVID19-2’, which contains data about the bivalent boosters. In this category, there are 38,360 reports of adverse reactions up to September 29, but 6,061 reports are listed as ‘no adverse event’.
A total 146,531 individual-symptom events are listed.
There are a total 353 reports of death after booster vaccination, but, in the ‘vaccine manufacturer’ category, there are 205 reports listed after administration of the Pfizer-BioNTech vaccine and 149 after administration of the Moderna vaccine (354 total).
There are 468 reports of permanent disability, but in the ‘vaccine manufacturer’ category, there are 284 reports listed after administration of the Pfizer-BioNTech vaccine and 185 after administration of the Moderna vaccine (469 total).
In the ‘COVID19-2’ category, there are 5,842 cases of Covid-19 and 5,340 reports separately listed as a positive SARS-CoV-2 test, 5,415 reports of a product storage error, 3,913 reports of an expired product being administered, 2,109 reports of the incorrect product formulation being administered, 1,699 reports of underdosing, 1,458 separate reports of an incorrect dose being administered, 1,028 reports of the wrong product being administered, 814 reports of an extra dose being administered, 469 reports of an inappropriate schedule of product administration, 460 reports of a “poor quality product” being administered, 423 reports of a product being administered to patient of an inappropriate age, and 335 reports of a “product preparation issue”.
ALL THE FOLLOWING DATA ARE FROM BOTH COVID VACCINE CATEGORIES COMBINED
There is now a disclaimer on the wonder.cdc.gov website that states the following: “At the request of European regulators, CDC and FDA have removed certain data fields (country codes; reported symptom case narrative free text; diagnostic laboratory data free text field; illness at time of vaccination free text field; chronic conditions free text medical history field; allergies free text field) from foreign VAERS reports which were submitted to VAERS and may not comply with European regulations. Domestic (U.S.) VAERS reports are not affected by this process.”
According to the latest data, VAERS received 18,197 reports of death after Covid vaccination from US states and territories or a location reported as unknown and 18,127 from foreign locations.
The 36,324 total includes the 205 reported deaths after administration of a Pfizer-BioNTech booster and 149 after administration of a Moderna booster.
According to VAERS, a total 22,917 deaths followed administration of the Pfizer-BioNTech vaccines, 10,531 followed administration of the Moderna vaccines, and 2,821 followed administration of the Janssen Biotech vaccine. In 263 cases, the name of the vaccine manufacturer was not specified in the report.
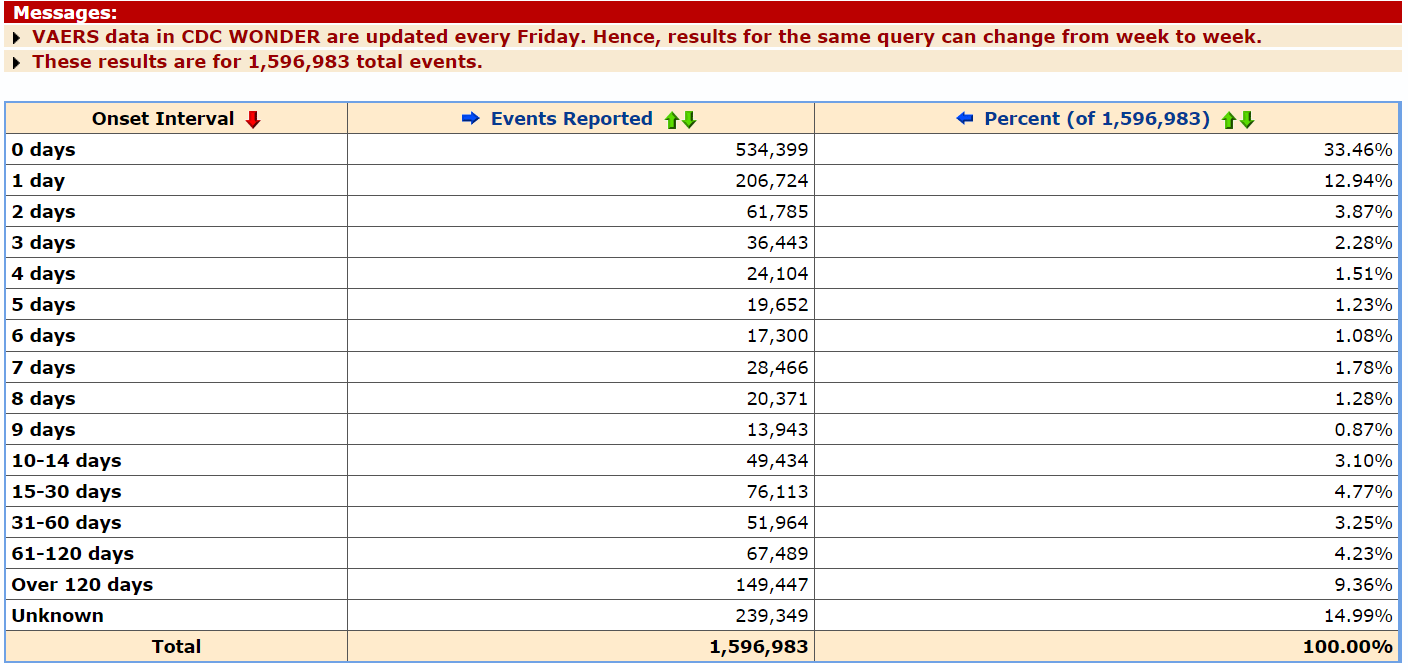
A total 4,218 of the reported deaths occurred on the same day as vaccination, 3,180 occurred the following day, 583 occurred within seven days, 1,637 occurred within ten to 14 days, and 2,820 occurred within 15 to 30 days.
According to Worldometers.info there had been 1,178,150 deaths from Covid-19 in the US as of October 6, 2023.
On VAERS 953,962 reports (up to September 29 and in all locations) relate to adverse events after administration of the Pfizer-BioNTech vaccines (4,576,117 individual-symptom events), 546,028 refer to the Moderna vaccines (2,286,767 individual-symptom events), 98,101 refer to the Janssen Biotech vaccine (445,210 individual-symptom events), 335 reports relate to the Novavax vaccine (1,482 individual-symptom events), and 13,077 relate to an unknown vaccine manufacturer (52,471 individual-symptom events).
The total number of reported adverse reactions resulting in permanent disability is put at 67,983. VAERS lists, as of September 29, 48,626 cases after administration of the Pfizer-BioNTech vaccines, 16,085 after administration of the Moderna vaccines, 3,358 after administration of the Janssen Biotech vaccine, and 12 after administration of the Novavax vaccine. In 405 cases, the vaccine manufacturer was not specified.
As of September 29 VAERS lists 11,574 reports of seizures after Covid vaccination. Some other adverse reactions such as generalised tonic-clonic seizure (1,313), seizure-like phenomena (654), and partial seizures (360) are listed separately.
There are 14,418 reports of a pulmonary embolism and 10,808 reports of thrombosis, with separate listings for specific types of thrombosis, such as deep vein thrombosis (9,479), pulmonary thrombosis (1,182) and cerebral venous sinus thrombosis (1,006). There are 212,981 reports of people being diagnosed with Covid-19 and 62,022 separate reports of a SARS-CoV-2 test being positive.
There are 10,047 reports of a cerebrovascular accident, 12,961 reports of sleep disorders, 14,250 reports of herpes zoster (plus 1,783 other herpes zoster reports listed separately, including herpes zoster reactivation), 2,463 reports of blindness, and 3,661 reports of deafness. As of September 29 there were 25,467 reports of tinnitus.
There are also 3,626 reports of spontaneous abortion (miscarriage) listed on VAERS as of September 29 along with 162 reports of stillbirth, 16,108 reports of heavy menstrual bleeding, 4,291 reports of intermenstrual bleeding, 9,517 reports of irregular menstruation, and 4,962 reports of delayed menstruation.
There are 3,422 reports of Guillain-Barré syndrome listed on VAERS as of September 29, along with 7,086 reports of Bell’s palsy, 9,460 reports categorised as facial paralysis, and 2,113 reports of facial paresis. There are also 231 cases of “facial asymmetry” listed on VAERS.
There are 17,185 reports of myocarditis (inflammation of the heart muscle) after Covid vaccination listed in the VAERS data up to September 29, 2023. VAERS lists 90 reports of viral myocarditis, 26 cases of eosinophilic myocarditis, 13 cases of giant cell myocarditis, nine cases of hypersensitivity myocarditis, nine cases of infectious myocarditis, six cases of immune-mediated myocarditis, six cases of chronic myocarditis, six reports of autoimmune myocarditis, four cases of septic myocarditis, three cases of post-infection myocarditis, three cases of mycotic myocarditis, two cases of bacterial myocarditis, two cases of coxsackie myocarditis, and one case of enterovirus myocarditis.
The VAERS data also includes 11,391 reports of pericarditis (inflammation of the tissue surrounding the heart), 783 cases of myopericarditis, 105 cases of pleuropericarditis, 64 cases of viral pericarditis, 41 cases of constrictive pericarditis, 16 cases of infective pericarditis, five cases of purulent pericarditis, three cases of adhesive pericarditis, three cases of rheumatic pericarditis, two cases of bacterial pericarditis, two cases of autoimmune pericarditis, one case of uraemic pericarditis, one case of gonococcal pericarditis, one case of cytomegalovirus pericarditis, and one case of lupus pericarditis.
A total 8,963 reports of an anaphylactic reaction and 1,548 reports of anaphylactic shock after Covid vaccination are listed up to September 29.
The CDC is no longer collecting adverse event reports on its v-safe app. It now directs those wishing to make a report about Covid vaccination to the VAERS website.
The CDC stated that it closed enrolment in v-safe for Covid-19 vaccines on May 19, 2023. V-safe, it noted, was developed specifically for Covid-19 vaccines.
“CDC is developing a new version of v-safe which will allow users to share their post-vaccination experiences with new vaccines,” the CDC added.
“CDC will continue to monitor the safety of Covid-19 vaccines through its other vaccine safety monitoring systems. V-safe users or others who get vaccinated can report any possible health problems or adverse events following vaccination to the Vaccine Adverse Event Reporting System (VAERS).”
There is more detail about the VAERS statistics on my Changing Times website along with data from other adverse event databases.
The World Health Organisation updates its VigiBase statistics every Sunday and the EudraVigilance statistics are usually updated on Mondays.